ID NOW™ COVID-19
MOLECULAR. IN MINUTES. ON THE FRONT LINE.
ID NOW™ is a leading molecular point-of-care platform in the United States, trusted by hospitals, physician offices, and urgent care clinics nationwide.
The ID NOW™ COVID-19 assay is now available for use on the ID NOW platform under U.S. Food and Drug Administration Emergency Use Authorization (EUA). The ID NOW™ COVID-19 rapid test delivers high-quality molecular positive results in as little as 5 minutes, targeting the coronavirus (COVID-19) RdRp Gene.
Timely results enable healthcare professionals to make appropriate and more efficient treatment and infection control decisions. EUA supports flexible near patient testing environments.
Only available in the US.
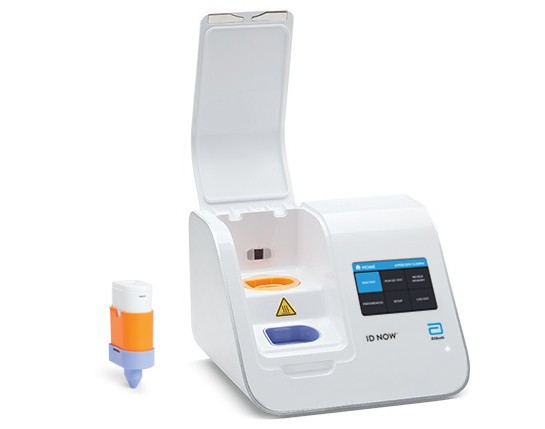

- Positive results may be detected in as little as 5 minutes
- Negative results in 13 minutes
- Molecular technology targeting COVID-19 RdRp gene
- Designed for near patient testing in a variety of healthcare environments1
- Room temperature storage
- Direct sample types include: Nasal, Throat, and Nasopharyngeal swabs
- Facilitates effective patient management
